|
|
Molecule of the Month October 2006 |
|
| Total Synthesis of Lepadiformine | ||
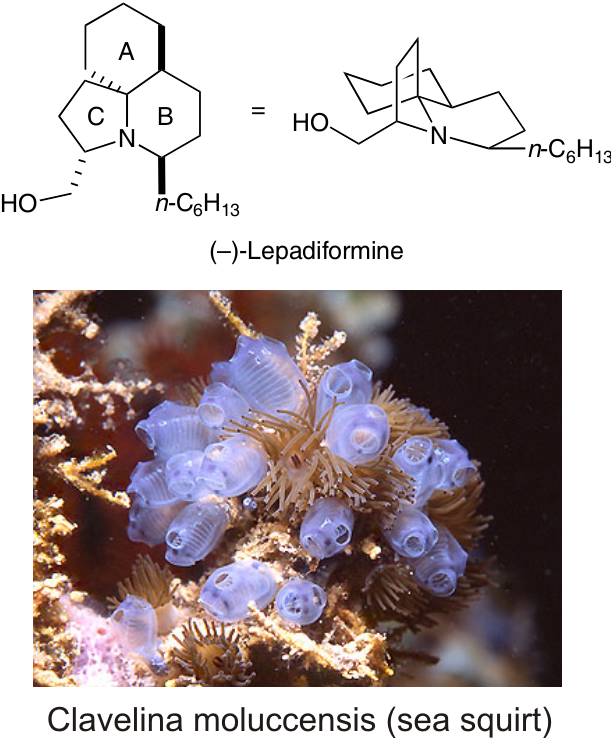
|
||
|
Lepadiformine is a marine alkaloid which was isolated in 1994 from the tunicate Clavelina lepadiformis and later from Clavelina moluccensis.
This work was carried out in the group of Prof. Philippe Renaud. References:
|
||